Introduction
Infectious diseases and their complications are a major cause of mortality, whose incidence decreased after the introduction of antimicrobial drugs in the 1940s, leading to an improved quality of life. Other public health measures that have contributed to better health status include water sanitation, immunizations, and the promotion of nutritious diets. The successful use of antibiotics has led to advances in medical-surgical techniques such as organ transplants, implant surgery, the care of premature newborns, and the treatment of immunocompromised patients, among others. 1
Antimicrobial resistance (AMR) is an emerging problem that threatens the effectiveness of these drugs, as it complicates the treatment of many infections. Since the widespread introduction of antibiotics and their indiscriminate use as a treatment regimen, bacterial resistance mechanisms have also evolved, becoming a significant public health challenge. 2 AMR is the ability of certain microorganisms to survive and multiply even when exposed to high concentrations of antibiotics that would normally be lethal. 3 To measure this phenomenon, scientists use the minimum inhibitory concentration (MIC) parameter, which indicates the minimum amount of antibiotic required to inhibit bacterial growth. However, some strains have developed the ability to withstand maximum concentration levels that would be lethal to other microorganisms of the same species. Bacteria can adapt, strengthen, and become difficult to eliminate through various mechanisms, such as intrinsic resistance, in which structural characteristics make them naturally tolerant to certain antibiotics; genetic mutations that develop over time; or horizontal gene transfer, through which they acquire resistance genes from chromosomal fragments or plasmids. 4 As resistance mechanisms multiply and combine, the treatment of infections becomes more complex, highlighting the urgency of understanding how these processes are evolving. 2
UTIs are among the most common infectious diseases worldwide, primarily affecting women, although they remain relatively under-researched. 5 In a study conducted among active-duty women in the Australian Defence Force, 41% of participants reported having experienced at least one UTI. UTIs and other genitourinary infections can impact the health and work performance of these women, emphasizing the importance of implementing prevention and management strategies within the military setting to reduce their impact. 6
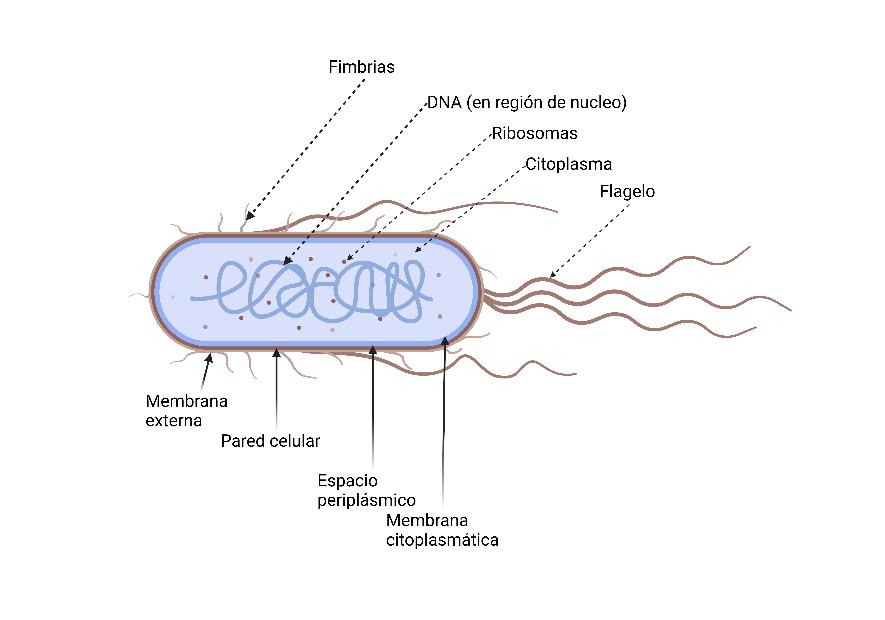
The most frequently isolated causative agent in UTIs is uropathogenic Escherichia coli ( Fig. 1 ) 7 . However, it is not the only species capable of colonizing the urinary tract ( Fig. 2 ) 8 . The interaction between pathogenic microorganisms, the host’s immune system, and the conditions present in the bladder determines the development and persistence of the infection. 5 A study conducted among university women who had experienced their first UTI episode revealed that 27% had at least one culture-confirmed recurrence within the following six months, and 2.7% experienced a second recurrence during the same period. When the initial causative agent is Escherichia coli, the likelihood of a new infection within the next six months is higher compared to other microorganisms. Another study involving women aged 17 to 82 with cystitis caused by E. coli showed that 44% experienced at least one recurrence within the following year. 9 , 10
UTI AND ETIOPATHOGENESIS
The bacteria responsible for recurrent urinary tract infections (rUTIs) originate from the gut microbiota and use virulence factors, such as pili or fimbriae, to colonize the urinary tract. The characteristics of the vaginal and bladder epithelium also influence susceptibility to bacterial colonization, either facilitating or hindering the adherence of uropathogenic bacteria (Table 1). 11
| Epithelium | Adhesion Mechanism | Factor | Description |
|---|---|---|---|
| Vaginal Cells | Colonization Propensity | Receptors for E. coli | Women with rUTIs have a higher number of E. coli receptors in the vaginal epithelium. |
| Genetic Factors | - HLA-A3 is associated with a higher risk of rUTIs. | ||
| - Women over 65 years old have more uropathogens adhered to the vaginal epithelium. | |||
| "Non-secretors" of ABO antigens | Women who do not secrete ABO antigens have a 3-4 times higher risk of rUTIs. Their epithelium facilitates uropathogen adhesion. | ||
| Vaginal Fluid | In non-colonized women, vaginal fluid inhibits bacterial adhesion, with secretory immunoglobulin A (IgA) playing a key role. | ||
| Bladder Cells | Bacterial Adhesion | E. coli Type 1 Pili | E. coli type 1 pili bind to uroplakins in bladder cells, facilitating colonization. |
| Bacterial Biofilms | Biofilm Formation | Bacteria form intracellular bacterial communities (IBC) similar to biofilms, allowing them to evade the immune response and persist in the urinary tract. It favors infection recurrence. | |
| Uromucoid (Tamm-Horsfall Protein) | Blocks adhesion of strains with type 1 pili. The absence of this protein increases E. coli colonization in animal models. |
Note: Created based on the article “The global burden of antimicrobial resistance-urinary tract infections”. 11
The etiology of UTIs is determined by identifying the microorganisms responsible for the infection, with their frequency varying according to age, sex, and the acquisition setting. In an analysis of 12,204 urine cultures, Escherichia coli was the most common pathogen, with a prevalence of 37–38% in adults and a higher incidence in children (59–64%). In adults, the other most frequent microorganisms were Enterococcus faecalis (16–18%) and Klebsiella pneumoniae (7–8%), while in children, E. faecalis reached 19% in community-acquired infections and 17% in hospital-acquired infections, followed by Proteus mirabilis (3–5%). Additionally, Candida albicans was the most prevalent fungus, with a frequency close to 5% in community infections and 4% in hospital infections. Concerning antibiotic susceptibility, no significant differences were found in the activity of antibiotics against E. coli in both adults and children, regardless of the infection acquisition setting. Similarly, in children E. faecalis and P. mirabilis showed no significant variations in antimicrobial susceptibility between community and hospital infections. 12
A study conducted with 457 children attending outpatient and emergency services at the Federico Gómez Children’s Hospital in Mexico, presenting uncomplicated lower urinary tract infection symptoms, identified the causative pathogens and evaluated their antimicrobial susceptibility. It obtained urine samples by midstream clean-catch or catheterization. The most frequently isolated pathogens were Escherichia coli (68.3%), Enterococcus spp. (11%), Klebsiella pneumoniae (8.7%), Pseudomonas aeruginosa (7.5%), Proteus mirabilis (4.5%), and Enterobacter cloacae (1.7%). 13
High resistance to various antibiotics was observed in some pathogens: resistance to trimethoprim/sulfamethoxazole was 73.7% in E. coli, 62.2% in K. pneumoniae, and 100% in P. aeruginosa, while resistance to ampicillin reached 86.3% in E. coli and 100% in P. aeruginosa. Regarding ciprofloxacin, resistance was lower, with only 33.8% of E. coli strains showing resistance. However, nitrofurantoin exhibited much lower resistance, especially among enterobacteria, with only 4.4% resistance in E. coli. These results highlight the high antimicrobial resistance in pathogens responsible for lower urinary tract infections in children, representing a significant challenge to adequately treating these infections. 13
Risk factors for UTIs
The higher predisposition of women to UTIs is largely due to their anatomy ( Fig. 2 ) 8 , such as the short length of the urethra, which facilitates the entry of pathogens into the bladder. 14
Besides behavioral, genetic, and urological factors also play relevant roles in the risk. Although some women are more prone to developing and maintaining recurrent UTIs, the reasons for this variability are not yet fully understood. It has been suggested that some Escherichia coli strains, by forming extracellular and intracellular biofilms, may contribute to the persistence of infections by releasing bacteria in a delayed manner within the bladder. 14
The risk factors for recurrent UTIs are similar to those for cystitis, linked to the frequency of sexual intercourse, personal history of urinary infections, family history of UTIs in the mother, and having experienced the first episode before the age of 15. Other influences include using spermicides, recent antibiotic administration, and medical conditions such as diabetes. In postmenopausal women, urinary incontinence, the presence of cystocele, post-void residual urine, and a history of gynecological surgery increase susceptibility to these infections. 15
Recurrent urinary tract infections (rUTIs) in women are a common condition that increases with age and usually occurs in patients without structural abnormalities of the urinary tract. Its origin is mainly due to vaginal colonization by uropathogens, facilitated by factors such as the use of spermicides, frequent sexual activity, maternal history of UTIs, and recent antimicrobial use. Despite its high prevalence, no association has been found between uncomplicated rUTIs and chronic kidney disease or hypertension. Prevention strategies include continuous or postcoital antimicrobial prophylaxis, the use of an oral vaccine, and estrogen replacement therapy in postmenopausal women. Additionally, treatments under development, such as vaginal vaccines and lactobacilli administration, have shown promising results. 15 However, other options like cranberry consumption or ascorbic acid have not demonstrated effectiveness preventing recurrence. In selected cases, patient self-administered antimicrobial therapy has proven to be an effective alternative for women with good adherence to medical treatment. 16
To evaluate women with rUTIs, a thorough medical history and physical examination are essential to identify possible risk factors and define specific studies. However, in most cases, recurrent infections are due to reinfections rather than anatomical or functional alterations of the urinary tract. Therefore, several studies question the usefulness of invasive procedures such as intravenous pyelography, voiding cystourethrography, and cystoscopy, as they often do not reveal abnormalities relevant to disease management. In the initial evaluation of patients without risk factors, non-invasive tests such as renal and pelvic ultrasound, measuring post-void residual volume and uroflowmetry, are prioritized. These studies allow the detection of alterations that justify referral to a specialist. 11
Recent research on the human urinary microbiome has enabled the development of specialized techniques to more precisely analyze a microbial community that is difficult to access and has low biomass. In the absence of UTIs, the concentration of microorganisms in urine is typically low, ranging from less than 100 to up to 10 , 5 colony-forming units (CFU) per milliliter. 17 This low microbial density requires particularly sensitive sampling and analysis methods, as highlighted by the study of Karstens et al. 18 Dysbiosis, understood as an imbalance in the microbial community, has been identified as a key factor in the predisposition to recurrent UTIs. The loss of protective commensal bacteria such as Lactobacillus and the increase of potentially pathogenic microorganisms like Escherichia coli promote colonization, perpetuate infection, and may contribute to the emergence of multidrug-resistant strains. This situation is exacerbated by the recurrent and non-specific use of antibiotics, which further disrupts the urinary ecosystem and perpetuates a vicious cycle of dysbiosis and infection. 18 , 19 , 20
ANTIMICROBIAL TREATMENT
Antibiotic treatment of an acute urinary tract infection (UTI) episode follows principles similar to those managing non-recurrent and uncomplicated UTIs. According to international guidelines, recommended first-line antibiotics include nitrofurantoin, fosfomycin trometamol, and pivmecillinam, while trimethoprim/sulfamethoxazole may be used in areas with E. coli resistance rates below 20%. Second-line options include cephalosporins. Treatment duration is usually short, generally between 1 and 5 days, depending on the antibiotic used. In mild cases without risk factors, physicians can consider a symptomatic management approach with increased fluid intake and the use of nonsteroidal anti-inflammatory drugs (NSAIDs) to reduce the need for antibiotics and minimize resistance development. 15
A study analyzed antimicrobial resistance in urinary tract infections among children over a five-year period. Resistance rates to various antibiotics were observed in bacteria isolated from urine samples of children with urinary infections. Among the key findings, a high prevalence of resistance to trimethoprim/sulfamethoxazole and ampicillin was found in common pathogens such as Escherichia coli, Klebsiella pneumoniae, and Pseudomonas aeruginosa. Additionally, resistance to last-line antibiotics such as ciprofloxacin and nitrofurantoin was notably low, suggesting that these options remain effective against some bacterial strains. This study underscores the importance of resistance rates and continuous monitoring to guide the treatment of urinary infections in children. It highlights the growing concern about antimicrobial resistance in common pediatric infections. 21
Goff et al. emphasize the urgent need for international and multidisciplinary collaboration to tackle antimicrobial resistance (AMR) by using antibiotics rationally through cooperation, mutual learning, and local adaptation. They also stress the importance of all healthcare professionals who prescribe antibiotics understanding the social impact of inappropriate use and adopting responsible practices. The publication provides concrete examples of how sharing experiences and effective policies can strengthen antimicrobial stewardship programs worldwide. 22
ANTIMICROBIAL RESISTANCE IN UTIs
The pathogens primarily linked to antimicrobial resistance (AMR) in urinary tract infections (UTIs) are gram-negative bacteria. Uropathogens have developed virulence and resistance mechanisms, indicating that these factors are often acquired together. The main resistance mechanisms studied in gram-negative bacteria include the production of extended-spectrum β-lactamases (ESBLs), which inactivate various β-lactam antibiotics; carbapenemases, capable of degrading carbapenems, considered last-line treatments; efflux pumps, which actively expel antibiotics from the bacterial cell; and porin modification, which reduces the entry of these drugs into the cell. 23 This resistance, especially in bacteria such as Staphylococcus aureus and Escherichia coli, has increased significantly over time, making eradication more complex and raising treatment costs. 24
MECHANISMS OF ANTIMICROBIAL RESISTANCE
In a study conducted in 2024, antimicrobial resistance in urinary tract infections (UTIs) was addressed, focusing on pathogens such as Escherichia coli, Klebsiella pneumoniae, Pseudomonas aeruginosa, and Enterococcus. The study analyzes various resistance mechanisms employed by these bacteria, including:
Production of β-lactamases: Enzymes that inactivate β-lactam antibiotics.
Modification of bacterial targets: Alterations in target proteins, such as penicillin-binding proteins and gyrases.
Changes in membrane permeability: Modifications in porins that restrict antibiotic entry.
Efflux pumps: Mechanisms that actively expel antibiotics from the bacterial cell.
Additionally, the article highlights the proliferation of E. coli strains that produce β-lactamase enzymes such as TEM and CTX-M, conferring resistance to first- and third-generation cephalosporins, as well as the rise of carbapenem-resistant strains—considered the last line of defense in severe infections. These findings underscore the need for new therapeutic strategies to combat antibiotic-resistant UTIs. 11 Among the main resistance mechanisms in gram-negative bacteria are the production of extended-spectrum β-lactamases (ESBLs), which inactivate various β-lactam antibiotics; carbapenemases, capable of degrading carbapenems—considered last-line treatments; efflux pumps, which actively remove antibiotics from the bacterial interior; and porin modifications, which reduce the entry of these drugs into the cell. These strategies enable bacteria to resist multiple treatments, complicating the management of UTIs and posing a significant challenge to healthcare systems worldwide. 23 A study analyzes the prevalence of antimicrobial resistance in Enterobacteriaceae isolated from the Peruvian Amazon region. The results reveal that a high percentage of Enterobacteriaceae strains are multidrug-resistant, with 57.4% of the isolates showing resistance to multiple antibiotics. Most of these strains produce extended-spectrum β-lactamases (ESBLs), with a notable prevalence of the bla_CTX-M gene. This situation highlights the growing concern over bacterial resistance in a region with limited medical resources, which complicates the treatment of severe infections. The study emphasizes the urgent need to implement effective control and prevention strategies to combat the spread of resistant strains, particularly in rural and hard-to-reach areas, where infections associated with these bacteria are more frequent. 25 Another study analyzes the prevalence of antimicrobial resistance in Escherichia coli isolated from clinical samples in hospitals in Peru, revealing growing concern over antibiotic resistance in the region. The results indicate that 57.4% of E. coli strains were multidrug-resistant, with a high prevalence of extended-spectrum β-lactamase (ESBL) producers, particularly the bla_CTX-M variants. This phenomenon highlights the urgent need for an appropriate control strategy against bacterial resistance, as therapeutic options are becoming significantly limited. The research also underscores the importance of epidemiological surveillance and the implementation of measures to prevent the spread of resistant strains in hospitals, especially in rural areas with limited medical resources. 26
Antibiotic resistance represents one of the biggest challenges in modern medicine, as it has evolved rapidly since the first reports of penicillin resistance in the 1940s. Over time, the extensive and indiscriminate use of these drugs has driven the emergence of multidrug-resistant (MDR), extensively drug-resistant (XDR), and, in the most critical cases, pan-resistant (PDR) bacteria—meaning that some infections no longer respond to any available treatment. 27 Resistance develops through specific mechanisms depending on the antibiotic structure and its mode of action, which include inhibition of cell wall synthesis, interference with DNA replication or protein synthesis, alteration of essential metabolic pathways, or disruption of the cytoplasmic membrane. However, bacteria have developed sophisticated strategies to counteract these effects, such as the production of enzymes that inactivate antibiotics, modification of their cellular mechanisms to prevent drug action, active expulsion of antimicrobial compounds via efflux pumps, and the transfer of resistance genes between different bacterial species, which accelerates the spread of the problem. In addition to biological and genetic factors, the misuse of antibiotics in human and veterinary medicine, self-medication, lack of adequate infection control measures in hospitals, and the use of antimicrobials in agriculture have significantly contributed to the rise in resistance. This global health crisis not only complicates the treatment of common infections but also endangers essential medical procedures such as surgeries, organ transplants, and cancer treatments, which rely on the effectiveness of antibiotics to prevent secondary infections. In this context, it is crucial to develop strategies that include the responsible use of antibiotics, research into new antimicrobial therapies, and the implementation of epidemiological surveillance policies that enable more effective control of the spread of resistant bacteria. 28
GENETIC TRANSFER MECHANISMS AND THEIR ROLE IN THE SPREAD OF ANTIBIOTIC RESISTANCE
Gene transfer plays a fundamental role in the spread of antimicrobial resistance (AMR), enabling bacteria to efficiently acquire and disseminate resistance genes. Among the genetic transfer mechanisms, horizontal gene transfer (HGT) is the primary pathway through which microorganisms exchange genetic information, facilitating rapid adaptation to hostile environments, such as those with high antibiotic pressure. The human gut microbiome functions as a vast reservoir of antibiotic resistance genes, allowing these genes to be transferred to opportunistic pathogens and contributing to developing resistant infections. The irrational and prolonged use of antibiotics in clinical settings has increased the abundance of these genes in the gut, enhancing their transmission. Studies have shown that certain antibiotics, such as levofloxacin, can induce genetic transformation in bacteria like Escherichia coli, further promoting the spread of resistance. Multiple mechanisms regulate gene transfer, including the restriction-modification system, which acts as a bacterial defense against the entry of foreign DNA by degrading it before it can integrate into the genome. On the other hand, the CRISPR-Cas system, known for its role in bacterial adaptive immunity, limits the spread of mobile genetic elements such as plasmids and phages that may carry resistance genes. It has been observed that multidrug-resistant bacteria, such as certain enterococci, often lack CRISPR-Cas, suggesting that this system could act as a natural barrier against the dissemination of resistance genes. However, some phages have evolved anti-CRISPR proteins that inhibit this system, enabling gene transfer even in bacteria with CRISPR-Cas defenses. These gene transfer processes, driven by the selective pressure of excessive antibiotic use, have significantly contributed to the antimicrobial resistance crisis. 29
NEW THERAPEUTIC STRATEGIES
Escherichia coli remains the primary etiological agent of community-acquired urinary tract infections (UTIs), showing high susceptibility to fosfomycin (96.5%) and nitrofurantoin (97.4%). However, resistance to commonly used antibiotics such as amoxicillin (58.1%), quinolones (36.6%), and trimethoprim-sulfamethoxazole (31.7%) limits their use in empirical treatment. These findings support using fosfomycin as a first-line treatment for uncomplicated cystitis in women under 65 years old and men under 15 years old, and nitrofurantoin is a suitable option for women under 65 years old. Given the observed resistance pattern, the empirical use of amoxicillin, amoxicillin-clavulanic acid, quinolones, and trimethoprim-sulfamethoxazole should be avoided in this clinical context. 30
According to Bader et al., first-line antimicrobial agents such as trimethoprim-sulfamethoxazole, nitrofurantoin, and fosfomycin remain viable options for uncomplicated UTIs. Their efficacy has declined due to the emergence of resistant strains, particularly extended-spectrum β-lactamase (ESBL)-producing Escherichia coli. 31 In these cases, carbapenems, such as ertapenem, have been widely used as the treatment of choice for complicated infections or those caused by multidrug-resistant pathogens. However, the overuse of carbapenems has raised concerns about the spread of resistance to this class of antibiotics, prompting the search for new therapeutic alternatives. In response to this challenge, new combinations of β-lactamase inhibitors with β-lactam antibiotics have been developed, demonstrating high efficacy against resistant pathogens. Among them, ceftolozane-tazobactam and ceftazidime-avibactam have shown significant activity against resistant Enterobacteriaceae, including ESBL-producing strains and some carbapenem-resistant isolates. Another promising agent is meropenem-vaborbactam, which combines a carbapenem with a potent β-lactamase inhibitor, providing coverage against carbapenem-resistant bacteria mediated by the KPC enzyme (Klebsiella pneumoniae carbapenemase). In addition to these combinations, new antibiotics with innovative mechanisms of action have been developed. Plazomicin, a next-generation aminoglycoside, has demonstrated efficacy against resistant Enterobacteriaceae, including those with aminoglycoside-modifying enzyme-mediated resistance. This agent is a viable option for treating complicated UTIs caused by multidrug-resistant organisms. Similarly, imipenem-relebactam has emerged as a promising therapeutic alternative for infections caused by multidrug-resistant pathogens by combining a carbapenem with a potent β-lactamase inhibitor.
The need for quick susceptibility testing and tailored antimicrobial therapy has been highlighted to enhance treatment effectiveness and minimize the emergence of resistance. Implementing molecular diagnostic tools allows for more precise identification of resistance mechanisms present in pathogens, facilitating the selection of the most appropriate antibiotic and minimizing unnecessary use of broad-spectrum antimicrobials. Additionally, strategies such as antibiotic de-escalation and monitoring antibiotic blood levels have proven effective in optimizing UTI treatment. 31
Advances in bacterial culture and DNA sequencing techniques have allowed the identification of a more diverse and previously unrecognized urinary tract microbiome (urobiome). This microbiome may play a crucial role in health and disease, particularly in urinary tract infections (UTIs), giving it significant clinical relevance. Although various studies have confirmed the existence of a core urobiome, the role of the microbes that compose it is not yet fully understood, especially concerning health and disease. In this article, we review the current state of research and conclude that the urobiome is an essential component of the body’s innate immune defenses and a valuable resource for developing new strategies for treating and controlling UTIs. 18 , 19 , 20
CONCLUSION
Genitourinary infections represent a global public health problem due to their high prevalence and increasing antimicrobial resistance. The overuse of antibiotics has favored the emergence of multidrug-resistant bacteria, complicating treatment and leading to higher therapeutic failures, hospitalizations, and costs. Among the resistance mechanisms, the production of extended-spectrum β-lactamases (ESBL) and horizontal gene transfer stand out. In this context, it is essential to promote the rational use of antibiotics, implement rapid susceptibility testing for timely diagnosis, and develop new therapeutic strategies and global surveillance policies to contain the spread of resistance genes in clinical and community settings.
References
- Peebles K Velloza J Balkus JE McClelland RS Barnabas RV High global burden and costs of bacterial vaginosis: a systematic review and meta-analysis. Sex. Transm. Dis. 2019 46 5 304 11
- Alós JI Resistencia bacteriana a los antibióticos: una crisis global. Enferm. Infecc. Microbiol. Clin. 2015 33 10 692 9
- Brauner A Fridman O Gefen O Balaban NQ Distinguishing between resistance, tolerance and persistence to antibiotic treatment. Nat. Rev. Microbiol. 2016 14 5 320 30
- Huemer M Mairpady Shambat S Brugger SD Zinkernagel AS Antibiotic resistance and persistence-implications for human health and treatment perspectives. EMBO Rep. 2020 21 12 e51034
- Murray BO Flores C Williams C Flusberg DA Marr EE Kwiatkowska KM Recurrent urinary tract infection: A mystery in search of better model systems. Front. Cell. Infect. Microbiol. 2021 11 691210
- O'Shea SD Pope R Freire K Orr R Gallagher N Genitourinary infections in Australian servicewomen. Neurourol. Urodyn. 2023 42 8 1668 75
- National Human Genome Research Institute Bacteria Internet 2024 cited 2025 Feb 17 https://www.genome.gov/es/genetics-glossary/Bacteria
- Instituto Nacional del Cáncer Infección urinaria Diccionario de cáncer 2024 cited 2025 Feb 17 https://www.cancer.gov/espanol/publicaciones/diccionarios/diccionario-cancer/def/infeccion-urinaria
- Alós JI Epidemiología y etiología de la infección urinaria comunitaria. Sensibilidad antimicrobiana de los principales patógenos y significado clínico de la resistencia. Enferm. Infecc. Microbiol. Clin. 2005 23 Suppl 1 3 8
- Ikäheimo R Siitonen A Heiskanen T Kärkkäinen U Kuosmanen P Lipponen P Recurrence of urinary tract infection in a primary care setting: analysis of a 1-year follow-up of 179 women. Clin. Infect. Dis. 1996 Jan 22 1 91 9
- Von Vietinghoff S Shevchuk O Dobrindt U Engel DR Jorch SK Kurts C The global burden of antimicrobial resistance–urinary tract infections. Nephrol. Dial. Transplant. 2024 39 4 581 8
- Artero-López J Gutiérrez-Soto B Expósito-Ruiz M Sorlózano-Puerto A Navarro-Marí JM Gutiérrez-Fernández J Etiología de las infecciones urinarias en nuestra área sanitaria y perfil de sensibilidad de los uropatógenos más frecuentes. Arch. Esp. Urol. 2021 74 2 97 207
- López-Martínez B Calderón-Jaimes E Olivar-López V Parra-Ortega I Alcázar-López V Castellanos-Cruz MDC Susceptibilidad antimicrobiana de microorganismos causantes de infección de vías urinarias bajas en un hospital pediátrico. Bol. Med. Hosp. Infant. Mex. 2014 71 6 339 45
- Zare M Vehreschild MJGT Wagenlehner F Management of uncomplicated recurrent urinary tract infections. BJU Int. 2022 129 6 668 78
- Valdevenito SJP Infección urinaria recurrente en la mujer. Rev. Chilena Infectol. 2008 25 4 268 76
- Valdevenito JP Álvarez D Infección urinaria recurrente en la mujer. Rev. Méd. Clínica Las Condes. 2018 29 2 222 31
- Rowe TA Juthani-Mehta M Urinary tract infection in older adults. Aging Health 2013 9 5 10.2217/ahe.13.38
- Karstens L Asquith M Caruso V Rosenbaum JT Fair DA Braun J Community profiling of the urinary microbiota: considerations for low-biomass samples. Nat. Rev. Urol. 2018 15 12 735 49
- Neugent ML Hulyalkar NV Nguyen VH Zimmern PE De Nisco NJ Advances in understanding the human urinary microbiome and its potential role in urinary tract infection. mBio 2020 11 2 e00218-20
- Jones J Murphy CP Sleator RD Culligan EP The urobiome, urinary tract infections, and the need for alternative therapeutics. Microb. Pathog. 2021 161 Pt B 105295
- Moriyama B Henning SA Neuhauser MM Danner RL Walsh TJ Continuous-infusion beta-lactam antibiotics during continuous venovenous hemofiltration for the treatment of resistant gram-negative bacteria. Ann. Pharmacother. 2009 43 7 1324 37
- Goff D Kullar R Goldstein E Gilchrist M Nathwani D Cheng A A global call from five countries to collaborate in antibiotic stewardship: united we succeed, divided we might fail. Lancet Infect. Dis. 2017 17 2 E56 63
- Lepe JA Martínez-Martínez L Puesta al día en Medicina Intensiva: infecciones graves por gramnegativos multirresistentes. Mecanismos de resistencia en bacterias gramnegativas. Medicina Intensiva 2022 46 7 392 402
- Castro-Orozco R Barreto-Maya AC Guzmán-Álvarez H Ortega-Quiroz RJ Benítez-Peña L Patrones de resistencia antimicrobiana en uropatógenos gramnegativos aislados de pacientes ambulatorios y hospitalizados Cartagena, 2005-2008. Rev. Salud Pública (Bogotá) 2010 12 6 1010 9
- León-Luna D Fajardo-Loyola A Yareta-Yareta J Burgos-Espejo A Peralta-Siesquen C Galarza-Pérez M Caracterización molecular de enterobacterias multirresistentes en dos departamentos de la selva peruana. Biomédica 2021 41 Sp. 2 180 7
- Guzmán M Salazar E Cordero V Castro A Villanueva A Rodulfo H Multirresistencia a medicamentos y factores de riesgo asociados con infecciones urinarias por Escherichia coli adquiridas en la comunidad, Venezuela. Biomedica 2019 39 s1 96 107
- Džidić S Šušković J Kos B Antibiotic resistance mechanisms in bacteria: Biochemical and genetic aspects. Food Technol. Biotechnol. 2008 46 1 11 21
- Pulingam T Parumasivam T Mohd Gazzali A Mohd Sulaiman A Chee JY Lakshmanan M Antimicrobial resistance: Prevalence, economic burden, mechanisms of resistance and strategies to overcome. Eur. J. Pharm. Sci. 2021 170 106103
- Tao S Chen H Li N Wang T Liang W The spread of antibiotic resistance genes in vivo model. Can. J. Infect. Dis. Med. Microbiol. 2022 2022 3348695
- Aguinaga A Gil-Setas A Mazón Ramos A Alvaro A García-Irure JJ Navascués A Infecciones del tracto urinario. Estudio de sensibilidad antimicrobiana en Navarra. An. Sist. Sanit. Navar. 2018 41 1 17 26
- Bader MS Loeb M Leto D Brooks AA Treatment of urinary tract infections in the era of antimicrobial resistance and new antimicrobial agents. Postgrad. Med. 2020 132 3 234 50
